|
11/29/2023 0 Comments Moderna covid vaccine after effects
The Johnson & Johnson and Oxford-AstraZeneca vaccines appear to be somewhat less effective than the Pfizer-BioNTech and Moderna vaccines, but they have similar safety profiles and reported side effects such as sore arm, fever, or chills. This vaccine has already been approved for use in the United Kingdom and several other countries, but the developers may not be ready to submit an EUA request to the FDA until this spring. Researchers at AstraZeneca and Oxford University have also developed a vaccine against COVID-19. 4, and the FDA’s Vaccines and Related Biological Products Advisory Committee is scheduled to discuss later this EUA this month. Johnson & Johnson submitted an EUA request for its vaccine on Feb. Miriam Smith, the chief of infectious disease at Long Island Jewish Forest Hills in Queens, New York, told Healthline.Īdditional COVID-19 vaccines may soon become available in the United States, including vaccines from Johnson & Johnson and Oxford-AstraZeneca. “The side effect profile for both vaccines was very favorable,” Dr. The trials have also found that both vaccines are safe and generally well tolerated. Trial data has shown that two doses of the Pfizer-BioNTech vaccine are 95 percent effective at preventing COVID-19, while two doses of the Moderna vaccine are 94 percent effective. 
These can appear as a lump in the armpit, which has worried some women who thought it could be a sign of breast cancer. Swollen lymph nodes have also been reported. Some vaccine recipients also developed short-lived flu-like symptoms, such as fatigue, headache, body aches, chills, and fever. The most common reported side effect following vaccination is pain at the injection site. Pfizer-BioNTech submitted data for people 16 years and older, while Moderna submitted findings for people 18 years and older. Safe vaccines have some mild side effectsīefore issuing EUAs for the Pfizer-BioNTech and Moderna COVID-19 vaccines, the FDA reviewed the available data from ongoing clinical trials. They will comb through the data methodically,” he said, “and are committed to seeing efficacy and safety demonstrated prior to issuing an. 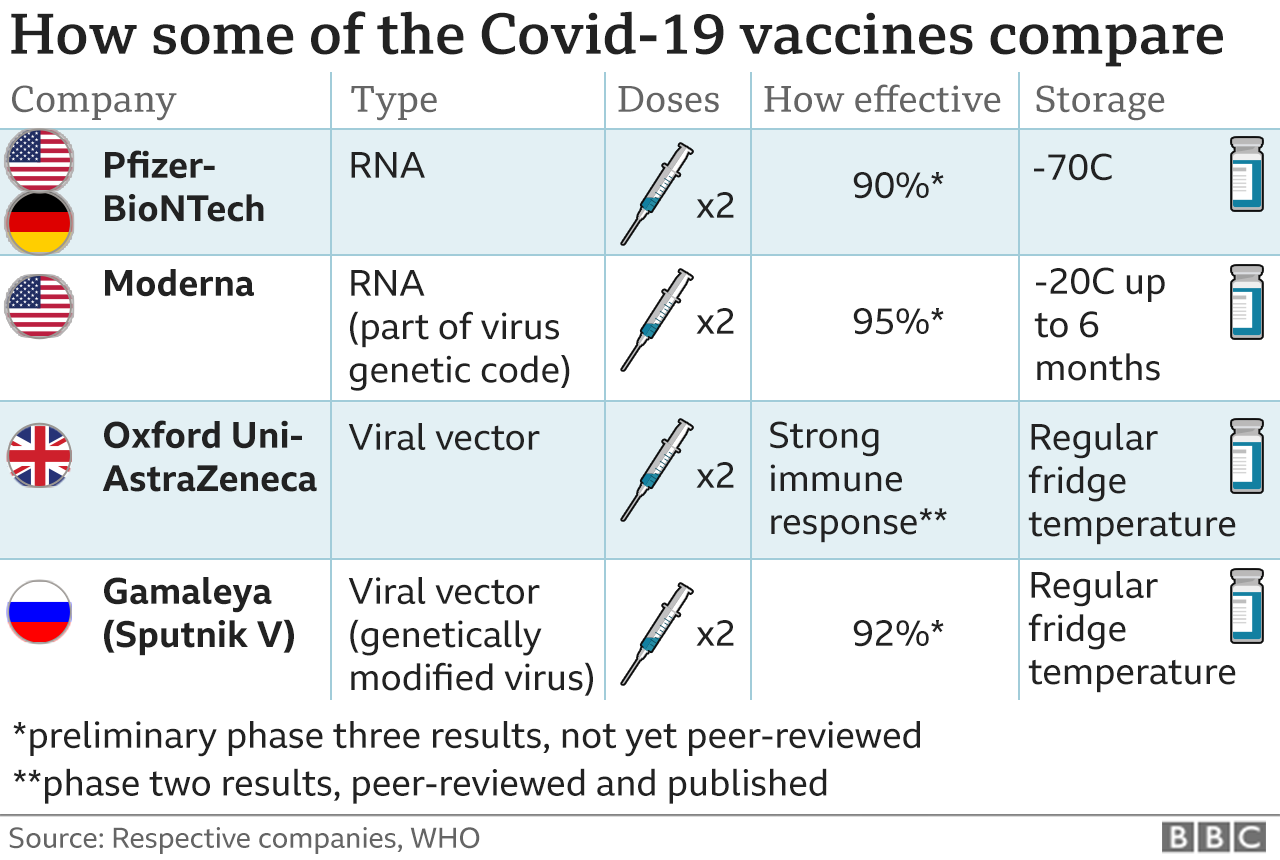
“But the FDA is extremely rigorous in their reviews. David Hirschwerk, an infectious disease specialist at Northwell Health in Manhasset, New York. “We are looking forward to it becoming available so that our vaccine supply can grow and synchronize with the demand,” said Dr. If the FDA finds that Johnson & Johnson’s vaccine is safe and effective enough to distribute, that will drastically increase the stock of vaccine doses in the country. In rare cases, severe allergic reactions have been reported, but in all those cases, people were successfully treated. Now the FDA is reviewing another EUA request for a vaccine developed by Johnson & Johnson’s subsidiary company, Janssen Biotech.Īll these vaccines appear to have mostly mild side effects that over-the-counter pain relievers can treat. In December 2020, the Food and Drug Administration (FDA) issued emergency use authorizations (EUAs) for two vaccines against COVID-19 - one produced by Pfizer and BioNTech, and one by Moderna.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
